Bioreactor processes traditionally rely on physical experimentation to tune parameters such as feeding strategies, agitation, and dissolved oxygen control to improve production. The complex nature of biologics production makes this approach slow, resource-intensive, and costly.
AI digital twins offer a new approach.
By combining real experimental data, a mechanistic understanding of biological systems, and predictive modeling, digital twins create a virtual representation of the bioreactor process.
This representation allows scientists and engineers to simulate process behavior, explore operating conditions, and optimize strategies before running physical experiments. Common biologics use cases include:
- Continuous Process Verification for batch monitoring and deviation detection
- Right first time scale up from lab to production
- Process optimization for production efficiency
The Bioreactor Digital Twin: Building a Process Model
Step 1: Making Bioreactor Data Model-Ready
The foundation of an accurate digital twin is process data, it’s what tailors the digital twin to your process. In biologics, datasets are often found in various formats: historian exports, spreadsheets, instrument files, and laboratory notebooks.
Typical examples of process data include:

This stage is about transforming the above data to ensure it can be properly fed into the model. For instance, time-series signals must be aligned, batch metadata must be preserved, and units must be reconciled across measurements such as nutrient concentration, gas flow rates, and agitation speed.
Proper data preparation ensures that the dynamic relationships between culture conditions and biological responses can be captured reliably within the digital twin.²
Step 2: Mechanistic Bioreactor Modeling
Mechanistic models are what ground your digital twin in scientific reality. Rather than relying solely on statistical correlations, these models describe process behavior using the underlying biological and physical principles governing cell culture.3
In bioreactors, this typically involves modeling cell growth kinetics, nutrient uptake, metabolite production, and product formation through systems of differential equations.
These models provide interpretability because model parameters correspond to real biological or engineering theories (i.e. growth rates, oxygen transfer coefficients, or yield coefficients). They also enable extrapolation, allowing predictions under operating conditions that have not yet been experimentally tested.4
However, mechanistic models alone cannot capture every aspect of biological complexity. Real-world bioprocesses exhibit variability and non-linear patterns that aren’t always captured by pure mechanistic models.
This is where hybrid modeling comes into play.
Step 3: Hybrid Modeling for Accurate Bioreactor Digital Twins
Hybrid modeling combines mechanistic models with data-driven learningdata-driven, learning to improve predictive performance while maintaining scientific plausibility.
The mechanistic component provides the biological and engineering foundation, ensuring and a Machine Learning (ML) layer is then used to capture underlying behaviors that are difficult to represent mechanistically.
For example, metabolic shifts during late-stage cultures or variability in nutrient consumption rates may not be fully captured by standard kinetic models. Data-driven corrections allow the digital twin to account for these effects using historical process observations.
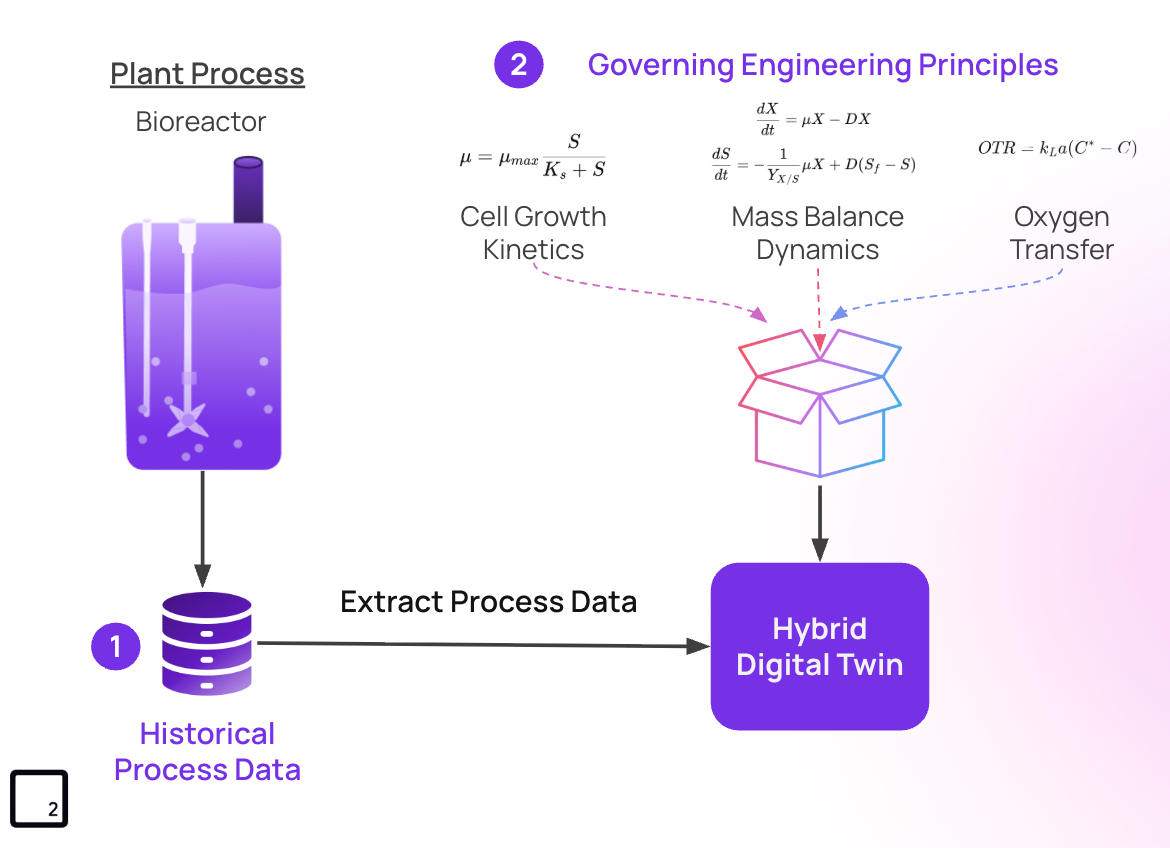
This enables a highly accurate bioreactor digital twin, providing a balance between scientific interpretability and predictive capabilities, which is particularly important in regulated biopharmaceutical environments.5
Step 4: In Silico Simulation for Bioreactors
Once validated, the digital twin becomes a powerful simulation environment.
Instead of executing large experimental campaigns, teams can gain visibility into their production and explore bioreactor operating strategies virtually.
This enables scientists to investigate questions such as how feed timing influences nutrient depletion, how agitation impacts oxygen transfer, or how temperature shifts affect titer. Virtual experiments allow researchers to systematically explore process design spaces that would otherwise require extensive laboratory work.
In silico experimentation therefore unlocks one of the most valuable capabilities of digital twins: the ability to evaluate a wide range of process conditions significantly faster than traditional experimentation.⁵
Stage 5: Your Bioreactor Digital Twin for Continuous Process Verification
Once a hybrid model accurately represents the process, engineers and operators can gain real-time visibility into how their process is run.
Here, they can visualize their critical quality attributes (CQAs) for each batch and compare them against historical performance, understanding any deviations in production, product quality, or titer.
The bioreactor digital twin can then be used in an environment that goes beyond understanding deviations to predicting them. By integrating the bioreactor digital twin with real-time data, operators can be notified when a deviation in temperature, for example, reduces the expected yield.
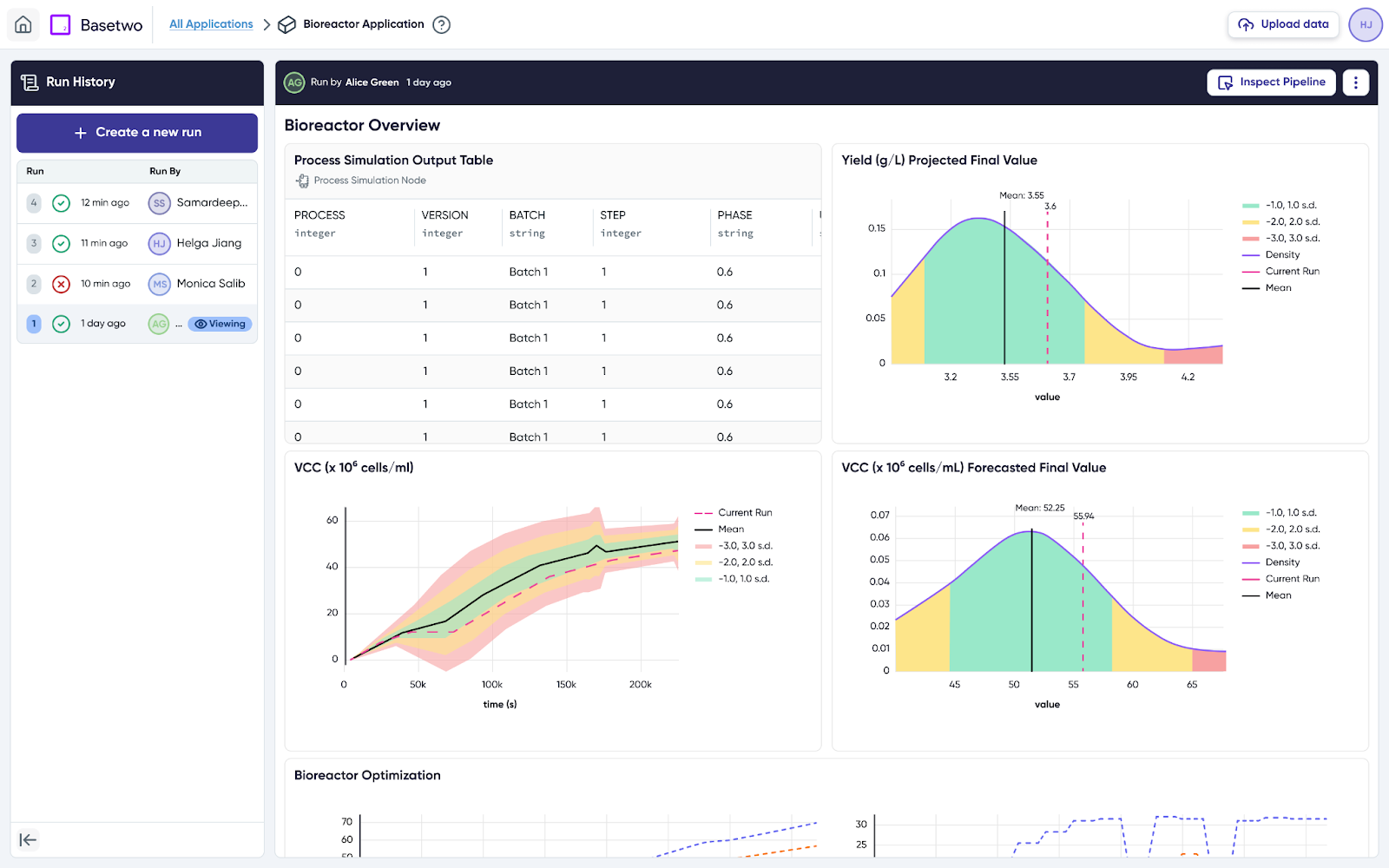
By moving from experimentation to real-time insights, digital twins enable a shift toward more data-driven and efficient process development.
How to Get Started with Bioreactor AI-Digital Twins
The value of digital twins in upstream bioprocessing is clear. However, many existing tools address only fragments of the lifecycle:
- Data systems without mechanistic engines
- Mechanistic simulators without lifecycle infrastructure
- ML models without physical grounding (hybrid capabilities)
Basetwo is the only platform that allows you to do every step:
Data integration → mechanistic modeling → hybrid calibration → simulation → process monitoring.
Rather than treating these steps as separate tools, Basetwo connects them all within one environment designed for bioprocess development.
The platform also links upstream and downstream operations, allowing teams to build digital twins that reflect the full manufacturing process rather than isolated unit operations.
If you want to improve your bio-ops with hybrid digital twins, Basetwo provides the infrastructure to make it possible: book a demo to learn more.



